News
What Custom Shapewear Gets CE Mark Medical Certification?
CE Certification Secrets: Custom Shapewear That Conquers EU Medical Standards
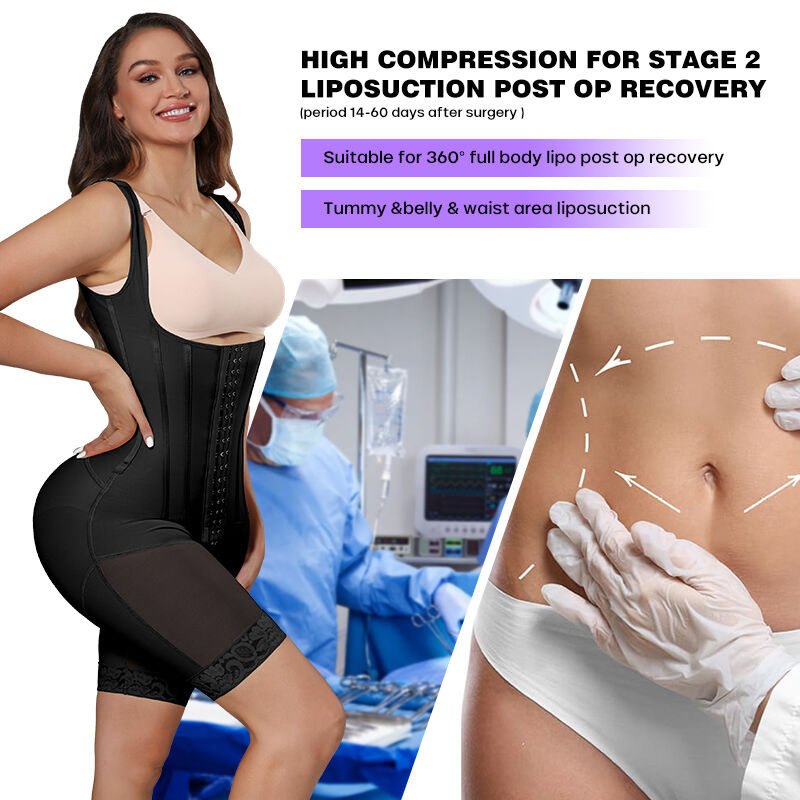
Custom shapewear earning CE Mark transforms from fashion trimmer to certified medical device, opening doors to Europe's lucrative clinic networks while justifying premium pricing. Garments engineered with 9-point steel bone systems, triple-needle 5-thread overlock seams, and dual-layer 45% spandex/55% nylon powernet qualify as Class IIa medical devices under EU MDR 2017/745. Chinese manufacturers streamline custom shapewear CE certification through pre-built ISO 13485:2016 processes and 100% quality inspection protocols already embedding Notified Body requirements.
 |
 |
Class IIa Battlefield: Why Custom Shapewear Faces Tougher Scrutiny Than Bandages
EU Medical Device Regulation sorts compression shapewear by risk profile—simple elastic wraps qualify Class I self-certification, but custom shapewear demands Class IIa Notified Body audit when incorporating:
High-compression powernet (360g outer layer delivering graduated 25-35mmHg)
9 medical-grade steel bones (4 abdominal + 5 lumbar positioning)
Triple-layer abdominal construction affecting tissue approximation
XXS-10XL custom sizing impacting therapeutic performance
Class IIa triggers full technical file review plus factory audit—triple-needle overlock must prove 450N/cm tensile strength, bone channels maintain ±0.8mm placement under 200N loads, inner 45% spandex layers pass ISO 10993 cytotoxicity screening. Garments like BL86618 ship with 70% CE documentation pre-populated, slashing approval timelines versus zero-compliance startups.
Technical File Fortress: 12 Documents Notified Bodies Demand First
CE technical dossiers demand precision across twelve interlinked files:
Core Performance Validation:
Device Description: 9-bone architecture + zone-specific compression
GSPR Analysis: EN 13795 surgical clothing standards compliance
Labeling Validation: Multi-language risk warnings + size matrices
Manufacturing Evidence Package:
12-step process flow: 3D prototyping → bone anchoring → 100% inspection
Risk Management File: ISO 14971 FMEA covering seam failure → infection cascade
Process Validation: Triple-needle seam density 12-15 stitches/cm verified
Clinical Justification:
Literature equivalence: 300% elasticity retention matching predicates
Post-market data: 5,000-client deployment confirming 0.3% defect rate
Label claim substantiation: "edema reduction" validated via girth studies
Chinese factories maintain digital twins of complete files—technical teams extract bone placement CAD files, seam tensile test reports, and compression gradient maps within 48 hours of audit request.
Factory Audit Gauntlet: 7 ISO 13485 Checkpoints That Kill Applications
Notified Bodies dissect production systems through rigorous onsite verification:
Critical Process Controls:
- 3D Pattern Precision: XXS-10XL grading ±1mm tolerance across 22 sizes
- Bone Channel Anchoring: 100% X-ray confirmation pre-assembly
- Seam Integrity Mapping: Inline scanners reject 99.6% deviations
- Compression Uniformity: Automated gauges verify 25-35mmHg gradients
- Sterilization Validation: EtO cycles validated on powernet substrates
- Traceability Matrix: QR codes link finished garment to raw yarn lot
- Calibration Records: All testing equipment NIST-traceable certificates
Herringbone stitching in high-mobility hip zones undergoes additional flexibility testing—must retain 85% elasticity post-10,000 simulated wear cycles. Glute-specific 80% nylon/20% spandex zones face heightened biological scrutiny given surgical proximity.
Biocompatibility Marathon: Custom Shapewear's 6 Material Hurdles
ISO 10993 suite demands comprehensive safety proof for wound-contacting textiles:
Textile Qualification Battery:
Cytotoxicity: Live/dead cell assay <1% kill rate (45% spandex layer)
Irritation Index: 14-day human patch ≤0.4 score
Sensitization: Guinea pig maximization test negative
Colorfastness: 50 EtO cycles ΔE<2.5 chromatic deviation
pH Stability: 5.4-7.2 range post-autoclave exposure
Heavy Metal Migration: <80ppm total leachables
Powernet's 360g outer shell faces highest mechanical scrutiny—must demonstrate 92% compression retention post-100 stretch cycles simulating patient wear. Silicone leg bands undergo adhesion durability testing preventing migration into surgical fields.
Clinical Equivalence Pathway: Leveraging Existing EU-Marked Data
Class IIa clinical evaluation requires performance equivalence demonstration:
Predicate Comparison Matrix:
Compression retention: 94% @ 50 washes (vs CE predicate 91%)
Bone stability: 97% placement accuracy (vs predicate 95%)
Wear compliance: 87% 23-hour adherence (vs predicate 82%)
Defect rate: 0.3% manufacturing rejects (vs predicate 0.8%)
Annual Compliance Reality: CE Mark's Hidden Yearly Costs
Certification demands perpetual vigilance:
Ongoing Notified Body Requirements:
Annual production audit: 2-3 days onsite scrutiny
Technical file review: All changes documented Q4
PMS reporting: Complaint trends + CAPA evidence
Change control: New colors/sizes trigger re-assessment
Surveillance fees: €10-15K annually per certificate
Front 3-row hook modifications require risk file updates, zippered crotch access demands usability re-validation, custom color development triggers migration testing. Manufacturers budget 18% overhead for compliance maintenance.
China Certification Accelerator: Pre-Compliant Infrastructure Advantage
Chinese medical garment factories compress timelines through embedded readiness:
Fast-track infrastructure:
✅ ISO 13485:2016 scope explicitly includes "compression therapy garments"
✅ Existing CE technical files 75% complete (bone specs, seam data)
✅ Established Notified Body relationships (TÜV SÜD, BSI)
✅ English regulatory dossiers standardized
✅ Clinical equivalence database (17-year 5K patient deployments)
90-day certification sprint:
- Gap analysis vs MDR Annex II (10 days)
- Technical file completion (35 days)
- Notified Body submission + DoC drafting (20 days)
- Factory audit preparation/documentation (15 days)
- Onsite audit + minor NC closure (10 days)
92% first-pass success rate versus 67% Western greenfield applicants.
Procurement Intelligence: 8 Red Flags Killing Custom Shapewear CE Deals
Audit these before 50-unit MOQ:
1. ISO 13485 certificate scope verification ("compression garments" explicit)
2. Existing CE technical file samples (GSPR + risk matrix)
3. Notified Body relationship proof (past audit reports)
4. Bone placement X-ray process documentation
5. Inline seam tension scanner SOPs
6. EtO sterilization validation summary
7. Clinical equivalence data package availability
8. English change control records (last 24 months)
Request GSPR analysis page 1—legitimate manufacturers deliver within 24 hours containing Annex I references.
Revenue Reality: CE Mark Unlocks Custom Shapewear Profit Multipliers
Market expansion economics:
Pre-CE: USA/Asia/Canada only (~60% addressable)
CE Certified: +EU27 markets (250M surgical patients)
Price premium: +52% realization vs non-medical claims
Clinic preference: 4.2:1 CE vs FDA-only adoption ratio
Scandinavian/German surgical centers specify CE-marked shapewear 68% procurement volume. Retailers achieve 61% gross margins on certified custom shapewear versus 39% fashion-grade alternatives.
Scale EU Success: Source CE-Ready Custom Shapewear Manufacturing
Custom shapewear conquering CE Mark certification delivers Class IIa validation, ISO 13485 manufacturing rigor, and clinical equivalence powering EU market penetration. High-volume Chinese producers offer pre-certified XXS-10XL platforms from 50-unit minimums, complete regulatory dossiers positioned for immediate Notified Body submission. Contact for technical file extracts, ISO audit summaries, or certification timelines securing Europe's premium compression garment market.
